MCQ Solution: Ionic Equilibrium
A solution contains a mixture of Ag+ (0.1M) and Hg22+ (0.1M) which are to be separated by selective precipitation. Calculate the maximum concentration of iodide ion at which one of them gets precipitated almost completely. What percentage of that metal ion is precipitated?
Ksp (AgI) = 8.5 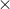 10–17
10–17
Ksp (Hg2I2) = 2.5 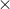 10–26
10–26
1) 90.83%
2) 97.83%
3) 89.93%
4) 99.83%
Solution:
Let us first calculate [I–] to precipitate AgI and Hg2I2
Ksp[AgI] = [Ag+] [I–]
8.5 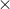 10–17 = (0.1) [I–]
10–17 = (0.1) [I–]
[I–] to precipitate as AgI = (8.5 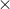 10-17)
10-17)
Ksp(Hg22+) = [Hg2I2][I–] = 8.5 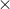 10–16 M
10–16 M
2.5 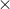 10–26 = 0.1 [I–]2
10–26 = 0.1 [I–]2
[I–] to precipitate Hg2I2 = 5.0 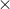 10–13 M
10–13 M
[I–] to precipitate AgI is smaller. Therefore, Ag I will start precipitating first. On further addition of I– more AgI will precipitate and when [I–] ³ 5.0 ´ 10–13 J, Mg2I2 will start precipitating. The maximum concentration of Ag+ at this stage will thus be calculated as:
Ksp(AgI) = [Ag+] [I–]
8.5 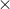 10–17 = [Ag+] (5.0 ´ 10–13)
10–17 = [Ag+] (5.0 ´ 10–13)
or, [Ag+] = 1.7 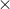 10–4 M
10–4 M
Percentage of Ag + remained precipitated = [(1.7 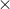 10–4 M)/0/1]
10–4 M)/0/1]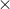 100 = 0.17%
100 = 0.17%
Thus percentage of Ag+ precipitated = 99.83%